Introduction: The Maillard Reaction as Coffee’s Flavor Engine
The transformation of green coffee beans into aromatic, flavorful roasted beans is a complex thermochemical process. Among the numerous reactions occurring during roasting, the Maillard reaction is the primary non-enzymatic browning pathway responsible for generating the vast majority of flavor and aroma compounds that define coffee’s sensory profile. This reaction, first described by Louis-Camille Maillard in 1912, is not unique to coffee; it is a cornerstone of flavor development in many thermally processed foods, including baked goods, roasted meats, and chocolate. However, in coffee roasting, its role is paramount, acting as the central chemical engine for creating flavor complexity. The reaction’s progression, governed by time and temperature parameters, directly dictates the sensory outcome, differentiating a light, acidic roast from a dark, bittersweet one. Understanding the Maillard reaction is therefore fundamental to the scientific control of roast profiles and the intentional design of coffee flavor.
Chemical Foundations: Amino Acids + Reducing Sugars = Flavor Complexity
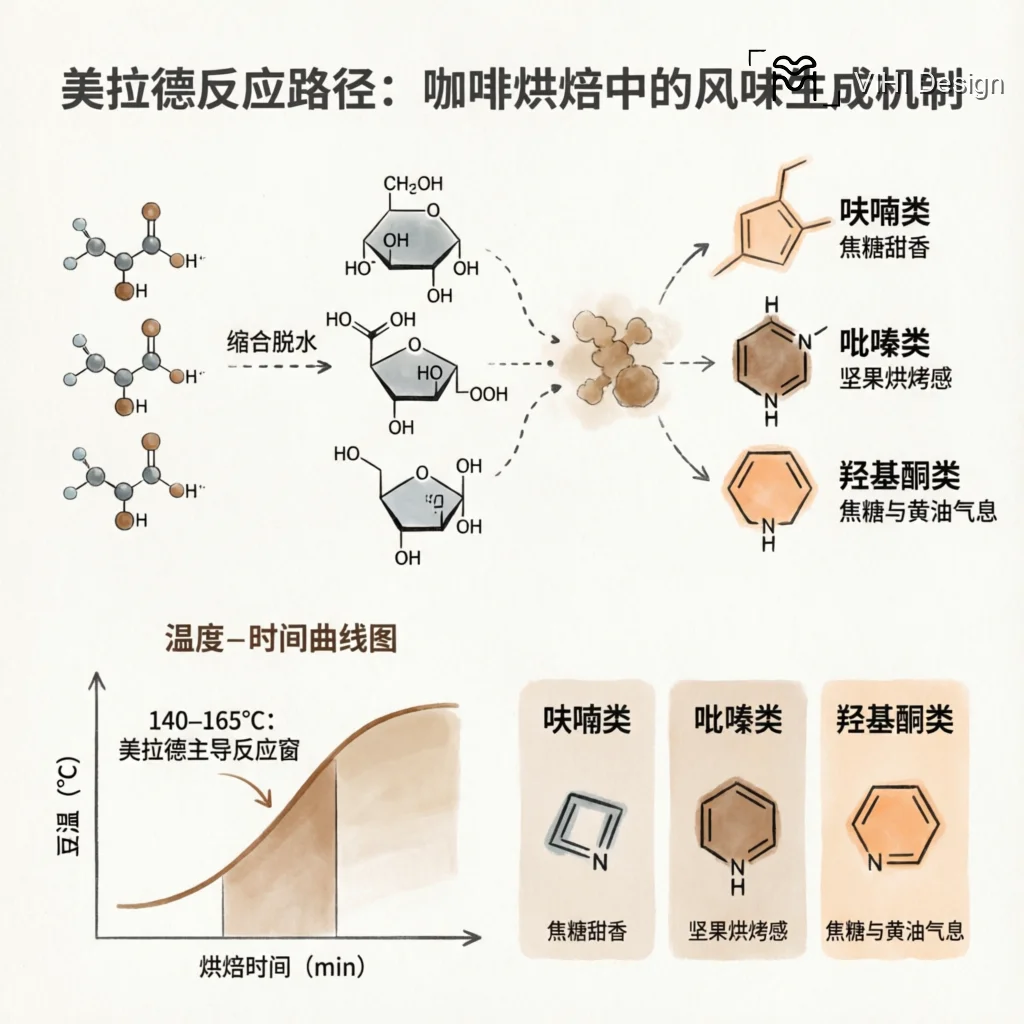
The Maillard reaction is a complex network of consecutive and parallel reactions between carbonyl groups, primarily from reducing sugars, and free amino groups, primarily from amino acids and proteins. Its chemistry can be broadly categorized into three stages: initial, intermediate, and final.
Initial Stage: Condensation and Rearrangement
The reaction initiates with a nucleophilic addition between the carbonyl group of a reducing sugar (e.g., glucose, fructose, arabinose prevalent in coffee) and the free amino group of an amino acid (e.g., asparagine, glutamine, lysine) or peptide. This forms an N-substituted glycosylamine, which undergoes an Amadori rearrangement (for aldose sugars) or Heyns rearrangement (for ketose sugars) to yield stable 1-amino-1-deoxy-2-ketoses or 2-amino-2-deoxy-aldoses, respectively. These Amadori or Heyns rearrangement products are key intermediates that dictate subsequent reaction pathways.
Intermediate Stage: Dehydration, Fragmentation, and Strecker Degradation
Under continued heating, the rearrangement products degrade via multiple pathways. Dehydration reactions produce furfurals (e.g., from pentoses) or hydroxymethylfurfural (HMF, from hexoses). Fragmentation reactions, such as retro-aldolization, create reactive dicarbonyl compounds like glyoxal, methylglyoxal, and diacetyl. A critically important parallel reaction is Strecker degradation. Here, α-dicarbonyl compounds from sugar fragmentation react with free amino acids. This results in the deamination and decarboxylation of the amino acid, producing an aldehyde (the Strecker aldehyde, e.g., methional from methionine, 2- and 3-methylbutanal from leucine/isoleucine) and an α-aminoketone. Strecker aldehydes are potent aroma compounds, while α-aminoketones are direct precursors to heterocyclic nitrogen compounds.
Final Stage: Polymerization and Heterocycle Formation
The reactive intermediates from the previous stage—furanones, pyrroles, dicarbonyls, Strecker aldehydes, and aminoketones—undergo polycondensation reactions to form high molecular weight, nitrogen-containing brown polymers known as melanoidins. Melanoidins contribute significantly to coffee’s color, body, mouthfeel, and antioxidant capacity. Concurrently, a vast array of key flavor-impacting heterocyclic compounds are formed through cyclization and condensation reactions. These include:
- Pyrazines: Formed from the condensation of α-aminoketones. Alkylpyrazines (e.g., 2,3-dimethylpyrazine, 2-ethyl-3,5-dimethylpyrazine) provide earthy, roasty, nutty, and cereal-like notes.
- Furans and Furanones: Derived primarily from sugar dehydration. Compounds like furfural (woody, bready), 5-methylfurfural, and sotolone (caramel, maple) contribute sweet, caramelic, and fruity aromas.
- Pyrroles and Pyridines: Formed from reactions involving carbonyls and ammonia or amino compounds. They often contribute roasted, earthy, and sometimes bitter notes.
- Thiophenes and Thiazoles: Formed when sulfur-containing amino acids (e.g., cysteine, methionine) participate in the reaction. These compounds are responsible for roasted, meaty, and sulfury notes at low concentrations.
Reaction Kinetics and Coffee-Specific Variables
The rate and extent of the Maillard reaction in coffee are non-linear functions of roast temperature and time. It typically becomes significant above approximately 150°C, with its rate increasing exponentially with temperature. The reaction’s progression is influenced by several coffee-specific substrate and process variables:
- Precursor Availability: The concentration and profile of reducing sugars and free amino acids in the green bean, which are influenced by coffee species (C. arabica vs. C. canephora), origin, and processing method (e.g., washed, natural, honey).
- Moisture Content: Water acts as a solvent and reactant medium. Its evaporation during roasting governs bean temperature kinetics and influences reaction pathways.
- pH: The reaction rate is generally favored under slightly alkaline conditions, but coffee’s inherent acidity modulates specific product formation.
- Thermal Profile: The precise time-temperature trajectory of the roast determines which stages of the Maillard reaction are emphasized, suppressed, or truncated, thereby shaping the final volatile and non-volatile compound profile.
In summary, the Maillard reaction is a cascade of chemical events initiated by the condensation of amino acids and reducing sugars. Its intermediate and final stages generate the complex mixture of volatile heterocyclic compounds and brown melanoidin polymers that collectively create the aroma, taste, color, and body of roasted coffee. Its kinetics and outcomes are the primary levers of control for the coffee roaster seeking to engineer specific flavor profiles.
Engineering the Maillard Reaction: A Technical Framework for Roasters
The roaster’s craft hinges on manipulating the Maillard reaction’s kinetic environment. We control three primary variables: thermal energy input, time, and the chemical state of the bean. The reaction rate roughly doubles for every 10°C increase in bean temperature, but this relationship is not linear. The availability of reactive precursors—specific amino acids and reducing sugars—diminishes as the reaction progresses, making the timing of heat application critical. A high initial rate of rise (RoR) can deplete these precursors prematurely, leading to a flat, simplistic flavor profile despite a fast roast.
Quantifying Outcomes: From Chemical Cascade to Brew Metrics
The success of Maillard management is ultimately measured in the cup. The compounds generated directly influence key brewing extraction metrics and sensory perception.
- Total Dissolved Solids (TDS 1.15% – 1.45%): Melanoidins contribute significantly to perceived body and viscosity. A roast with extended Maillard development typically yields a higher TDS at the same extraction yield, signaling greater solubility of these mid-weight polymers.
- Extraction Yield (EY 18% – 22%): The Maillard reaction creates both soluble compounds (acids, sugars, flavor molecules) and less-soluble polymers. Optimizing Maillard kinetics aims to maximize the extraction of desirable flavors before extracting excessive bitter compounds from the melanoidin backbone.
- Particle Size & Grind: The bean’s cellular structure, modified by Maillard-driven pyrolysis, determines fracture mechanics. A well-developed roast produces a more uniform particle size distribution when ground, leading to even extraction. Under-developed beans often shatter unpredictably, creating fines that cause astringency.
Barista’s Field Notes: Addressing Common Struggles
Translating theory into practice reveals consistent challenges. Here are direct interventions for common Maillard-related issues.
- Baked Flavors (Flat, Grainy, Bread-like): This stems from insufficient thermal momentum during the Maillard stage. The bean temperature plateaus, stalling the reaction cascade. Solution: Ensure a minimum bean temperature RoR of 5°C/min throughout the post-dry phase. Avoid drastic heat reductions before first crack.
- Confusing Maillard with Caramelization: Caramelization of sucrose occurs at much higher temperatures (>160°C) and is a minor player compared to the Maillard cascade. Mis-timing the “yellowing phase” as caramelization causes roasters to apply heat too aggressively, scorching sugars instead of fostering complex amino-sugar interactions.
- Identifying Maillard-Derived Flavors: In tasting notes, look for savory, nutty (almond, peanut), chocolaty (dark chocolate, cocoa), or malty descriptors. These are hallmarks of pyrazines and other Maillard compounds. Bright fruit notes typically originate from acids preserved earlier in the roast.
- Acidity Without Complexity: A roast that rushes to first crack preserves acidity but truncates Maillard development. The cup tastes sharp and simple. Pro-Tip: For maximum Maillard complexity without bitterness, extend the time between first crack and color change completion by 30-45 seconds while maintaining a bean temperature increase of 5-8°C per minute. This ‘Maillard window’ is where most flavor magic happens.
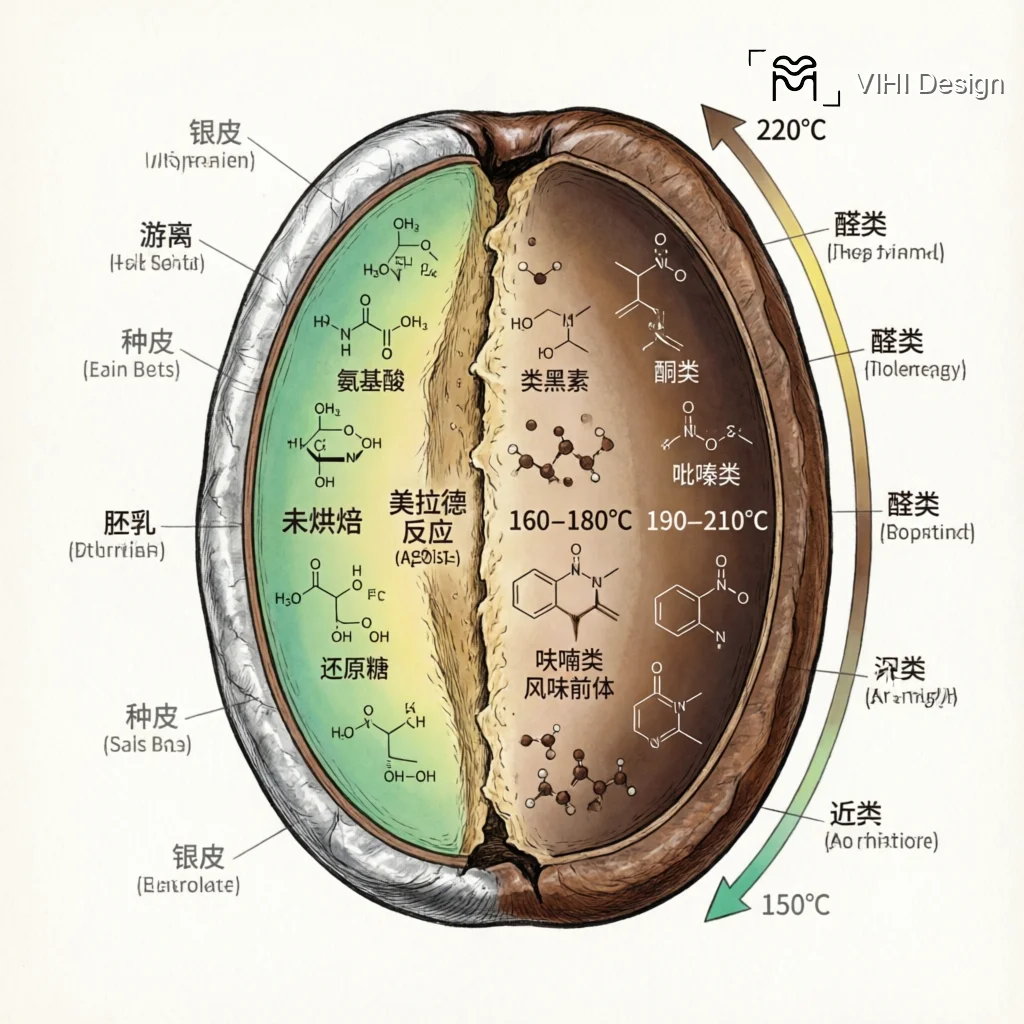
The Flavor-Development Continuum: A Stage-Based Model
Viewing the roast as a series of chemical thresholds, rather than just color change, refines profile design.
- Stage 1: Initiation (150°C – 160°C): Strecker degradation begins, producing aldehydes and nitrogen compounds. Aromas shift from grassy to toasted grain.
- Stage 2: Proliferation (160°C – 180°C): Exponential formation of pyrazines (nutty), thiophenes (sulfurous, roasted), and furans (caramel-sweet). This stage builds foundational flavor complexity.
- Stage 3: Polymerization & Diversification (180°C+): Melanoidin formation accelerates, contributing to body and color. Concurrently, secondary reactions create more complex heterocycles like alkylpyridines (earthy, bitter) that define roast depth.
Technical Summary
- The Maillard reaction is a non-enzymatic browning process between amino acids and reducing sugars, responsible for the majority of coffee’s aroma, flavor, color, and body.
- Its kinetics are primarily controlled by bean temperature and time; optimal development requires maintaining thermal momentum (5-8°C/min) through the critical 150°C-180°C window.
- Key sensory outputs include nutty, chocolaty, and malty flavors from compounds like pyrazines, with melanoidins directly influencing brew body and TDS.
- Common roasting errors—baked flavors, simplistic acidity—result from stalling the reaction or truncating its timeline before first crack.
- Precise manipulation of the post-first crack “Maillard window” (extending by 30-45 seconds) is a proven method for maximizing complexity without introducing harsh bitterness.
“`json
“`
“`json
“`
“`json
“`

Leave a Reply