Controlling Anaerobic Fermentation: pH, Temperature, and Gas Variables
Introduction to Anaerobic Fermentation as a Controlled Process
The application of anaerobic fermentation in coffee processing represents a significant shift from traditional post-harvest methods toward a more controlled, bioreactor-like environment. Initially popularized for its potential to produce distinct sensory profiles, the technique has often been discussed in marketing terms, emphasizing novelty and exotic flavor notes. However, its evolution into a legitimate tool for quality development necessitates moving beyond this narrative to establish a foundation of scientific precision. Anaerobic fermentation is defined here as the microbial metabolism of coffee cherry or mucilage substrates in an environment with severely restricted oxygen (O₂) availability. This oxygen limitation fundamentally alters the biochemical pathways available to microorganisms, shifting metabolism from oxidative to fermentative processes, with direct consequences for the chemical composition of the bean.
The core objective of controlled anaerobic fermentation is not merely the creation of an oxygen-free space, but the deliberate management of key process variables to guide microbial succession and metabolic output toward desired sensory outcomes. Uncontrolled fermentation, even in an anaerobic setting, risks the proliferation of spoilage microorganisms, the production of off-flavors, and batch inconsistency. Therefore, precision control transforms the fermentation tank from a simple container into a defined ecological niche. This control is exercised primarily through the modulation of three interdependent variables: the hydrogen ion concentration (pH), temperature, and the composition of the gaseous atmosphere. By manipulating these parameters, the processor can influence the rate of fermentation, the dominant microbial consortia, and the specific organic acids, alcohols, and esters produced, thereby directly shaping the acidity, body, and aromatic profile of the final cup.
Understanding pH Variables in Anaerobic Fermentation
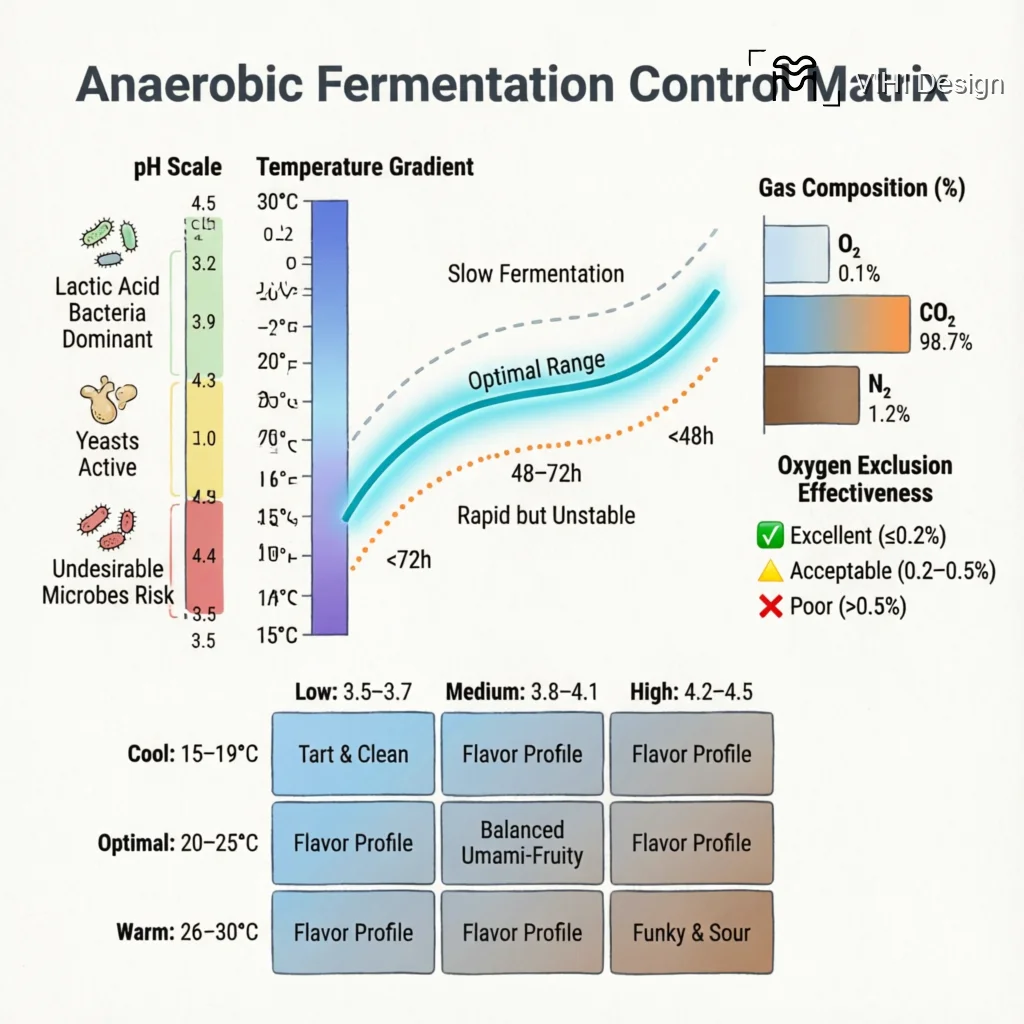
pH is a critical master variable in any fermentation process, serving as both an indicator of metabolic activity and a selective pressure that shapes the microbial ecosystem. In the context of coffee anaerobic fermentation, the typical operational range is between pH 3.5 and 4.5. This acidic environment is not a starting condition but a result of microbial metabolism, primarily the production of organic acids such as lactic, acetic, and citric acid. The dynamic change in pH throughout the fermentation period provides a real-time biochemical signature of the process.
pH as a Selective Agent for Microbial Activity
The progression of pH from a higher initial value (often near 5.0-6.0 in fresh mucilage) down to the 3.5-4.5 range is a key driver of microbial succession. Different microbial groups have distinct pH optima and tolerances:
- Initial Phase (pH >4.5): A broader community, including enterobacteria and some yeasts, may be active. As acid-producing bacteria (e.g., lactic acid bacteria, LAB) begin to metabolize sugars, pH drops.
- Mid-Fermentation (pH 4.0-4.5): This range becomes selective for acid-tolerant microorganisms. Lactic acid bacteria (e.g., Lactobacillus, Leuconostoc spp.) thrive and dominate, efficiently converting sugars into lactic acid, further acidifying the substrate. Certain yeasts, such as some Saccharomyces and non-Saccharomyces strains, remain active in this range.
- Low pH Stabilization (pH <4.0): A highly selective environment where only the most acid-resistant microbes persist. This can include specific strains of LAB and yeasts. This low pH acts as a biocontrol, suppressing the growth of many spoilage bacteria and fungi that could produce undesirable metabolites.
By setting a target terminal pH or monitoring the rate of pH decline, processors can indirectly steer which microbial populations become dominant, thereby influencing the acid profile of the fermentation.
pH Influence on Enzyme Activity and Bean Biochemistry
The internal environment of the coffee bean is influenced by the external fermentation pH. A lower pH in the mucilage can affect the activity of endogenous bean enzymes, such as pectinases and proteases, which are involved in the breakdown of mucilage and the modification of bean constituents. Furthermore, pH governs the ionization state of organic acids and other compounds. For instance, at a lower pH, a higher proportion of organic acids exist in their undissociated, lipophilic form, which may more easily diffuse across the parchment and seed coat into the bean. This direct incorporation of acids from the fermentation environment is a hypothesized mechanism for the development of specific acidic notes in the final cup. The precise relationship between fermentation pH, acid diffusion, and perceived sensory acidity requires further elucidation but remains a central tenet of process control.
pH as a Control Point and Termination Signal
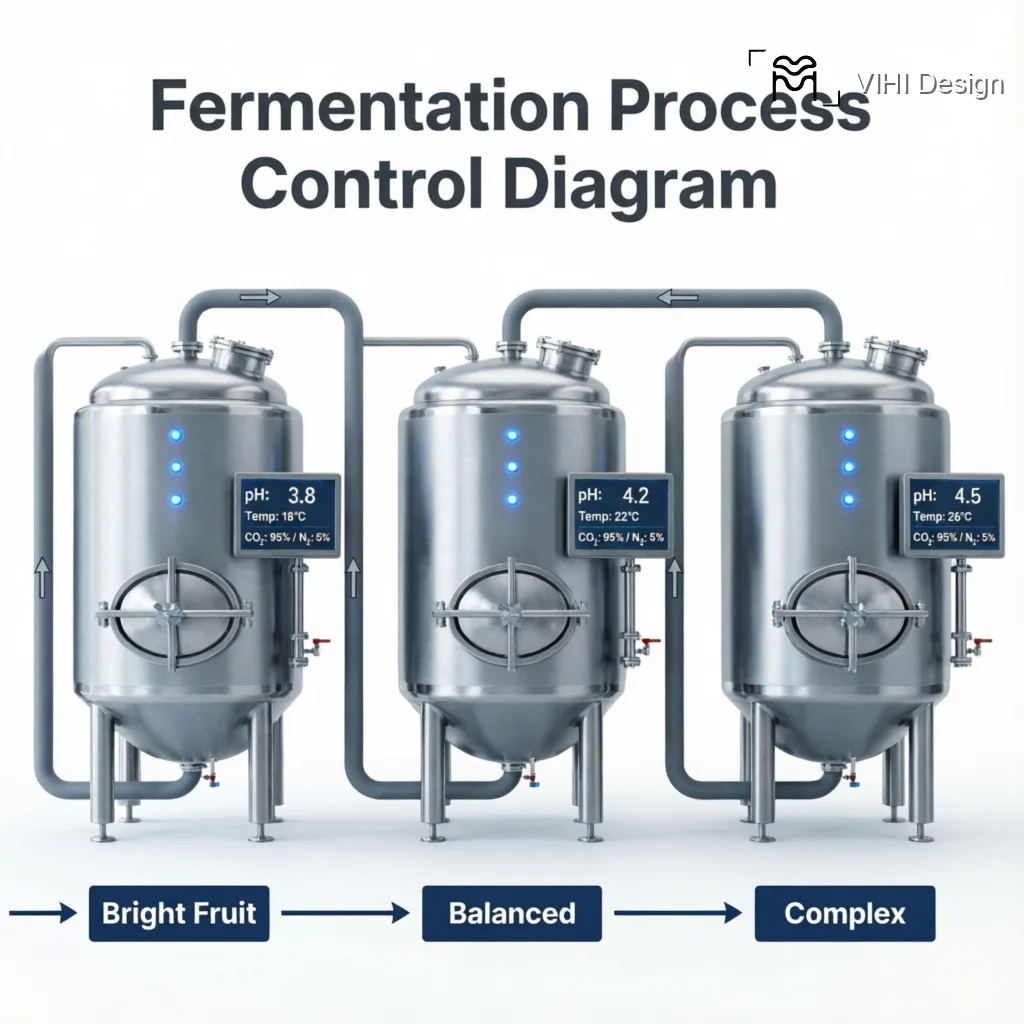
In a controlled process, pH is not a passive observation but an active control point. The rate of pH drop can be modulated by temperature (slower at lower temperatures) and initial substrate conditions. A rapid pH drop may indicate a highly active, homolactic fermentation, potentially yielding a cleaner, more pronounced lactic acidity. A slower decline may allow for a more complex succession of microbes. Crucially, reaching a target pH value (e.g., 4.0) is often used as a reliable, objective signal to terminate fermentation, as it indicates a specific level of metabolic conversion and microbial stability, enhancing batch-to-batch consistency compared to using time alone as a metric.
Interpreting pH Kinetics: From Data to Flavor Profile
Monitoring the pH drop rate provides a kinetic map of microbial activity. A rapid initial descent, or “H drop,” strongly suggests a dominant, fast-acting homolactic fermentation. This environment favors Lactobacillus species, which efficiently convert sugars into lactic acid. In the cup, we anticipate a cleaner, more singular lactic acidity—think of the bright, sharp tang of yogurt or ripe green apple. This profile often presents with remarkable clarity but may lack layered complexity if the microbial succession is truncated.
Conversely, a slower, more staggered pH decline indicates a more ecologically complex fermentation. This gradual acidification allows for a succession of microbial communities—yeasts, heterofermentative bacteria, and other acid-tolerant species—to contribute metabolites before the environment becomes too hostile. This process builds a broader spectrum of organic acids, esters, and higher alcohols. The resulting cup typically exhibits a more intricate, wine-like complexity, with layered fruit and sometimes floral notes, though it carries a higher risk of developing off-flavors if not meticulously managed.
Termination Protocols: The Science of the “Stop Signal”
Using time alone as a fermentation metric is fundamentally flawed due to variable ambient temperatures, differing microbial populations, and unique coffee cherry biochemistry. A target pH value, such as 4.0, serves as a robust, objective termination signal. This specific acidity level achieves several critical goals:
- Microbial Inhibition: A pH of 4.0 effectively halts the metabolic activity of most spoilage bacteria and fungi, stabilizing the batch.
- Metabolic Snapshot: It represents a specific, reproducible point in the sugar-to-acid conversion process, directly linking chemical state to sensory outcome.
- Consistency Engine: This method decouples the process from unpredictable environmental time, enabling producers to achieve remarkable batch-to-batch consistency regardless of minor temperature fluctuations.
This protocol transforms fermentation from an artisanal guess into a repeatable scientific process, providing a tangible lever to control quality.
Barista’s Field Notes: Addressing Common Struggles
Across the supply chain, professionals encounter specific, recurring challenges with processed coffees. Here is a breakdown of these pain points from a technical perspective.
- Producers & Inconsistency: Simply sealing a tank does not guarantee a consistent anaerobic environment. Variables like cherry temperature, Brix level at harvest, and native microflora create vastly different starting conditions. Without monitoring pH kinetics, each batch follows an unknown metabolic path, leading to unpredictable results despite the “anaerobic” label.
- Roasters & Unpleasant Surprises: Roasters often receive lots with jarring, vinegar-like acidity or muted, overly funky profiles. This usually stems from unchecked heterofermentative activity (producing acetic acid) or, conversely, a fermentation that was stopped too early or too late, failing to develop a stable, pleasant acidity.
- Farmers & Equipment Access: High-precision lab pH meters are cost-prohibitive for many. However, affordable, calibrated handheld meters with 0.1 pH resolution are now viable for field use. The barrier is often knowledge, not just technology.
Pro-Tip: When cupping anaerobic coffees, pay attention to the ‘fermentation window’ – the narrow temperature range (20-22°C) where pH control becomes most critical for developing clean, complex fruit notes without vinegar-like acidity. Coffees fermented outside this range often require more aggressive pH management to achieve balance.
Optimizing Extraction: Bridging Process to Brew
The unique soluble compound profile of a meticulously fermented coffee demands tailored extraction. The pronounced organic acid content and altered sugar complexes interact with water differently than washed coffees.
- Total Dissolved Solids (TDS): Target a slightly lower range of 1.15% – 1.45%. The heightened perceptibility of acids and esters means over-extraction (bitter, astringent) occurs at a lower TDS than with traditional profiles.
- Extraction Yield (EY): Aim for 18% – 22%. Staying within this window maximizes the positive fruit and acid complexity while minimizing the risk of extracting harsh phenolic compounds.
- Particle Size: Use a moderately coarser grind than for a washed coffee of the same roast level. This slows the extraction rate, providing more control to highlight the delicate, often volatile, aromatic compounds before pulling out bitterness.
Adjust your brew recipe with these parameters as your baseline, letting the distinct fermentation signature guide your final adjustments.
Technical Summary
- A rapid pH “H drop” typically indicates homolactic dominance, yielding a cleaner, more pointed lactic acidity.
- A slower, staged pH decline suggests complex microbial succession, potentially creating a more intricate, wine-like cup profile.
- Terminating fermentation at a target pH (e.g., 4.0) provides an objective, metabolic-based “stop signal” that dramatically improves batch consistency over time-based methods.
- For optimal brewing, target a TDS of 1.15%-1.45%, an EY of 18%-22%, and a moderately coarser particle size to best express clean fermentation characteristics.
- The critical fermentation temperature window for clean fruit development is 20-22°C, where precise pH management is most effective at preventing off-flavors.
“`json
“`

Leave a Reply